When only very small amounts of material are available for use during early stages of development, obtaining valuable, high-quality results is essential for progressing development of lead candidates. The amount of material required for these small-scale microscopical screens can range from nanogram to low microgram quantities, depending on the specific drug substance. Three different screening techniques can be employed for studying polymorphs, salts, and/or cocrystals including: 1) vapor crystallization within a small, closed glass chamber, 2) microscope well-plate, or 3) thin film. Each of these techniques can mimic traditional larger scale screening studies while using only a small fraction of the material typically required, ultimately increasing the number of relevant experiments that can be carried out. In addition, a variety of solvents, solvent mixtures, cooling rates and concentration gradients can be used to mimic potential process conditions or to find the presence of metastable forms.
The unique advantage of microscreen studies allows the material to be exposed to appropriate conditions on a specific slide and characterized directly by FT-IR and Raman microscopy without the need to transfer the material for analysis. In addition, crystalline materials obtained from these studies can then be isolated, scaled-up as needed, and further characterized using appropriate physical characterization techniques including: UV/VIS and NMR spectroscopy, thermal analysis by DSC, TG and hot-stage microscopy, and XRPD. Ultimately, the crystal structure could be solved if suitable single crystals are grown.
Combining the expertise of Improved Pharma’s solid-state chemists and microscopists enables studying solid forms of materials using nanogram to microgram quantities of material. These micro-scale screens can provide valuable insight for progressing the development of lead compounds when only a minimal amount of material is available. Solid-state screening studies can be carried out to select the appropriate candidates during earlier stages of development, thereby shortening the overall drug development timeline.
To learn more about Improved Pharma’s approach to polymorph and salt screening, please refer to this presentation.
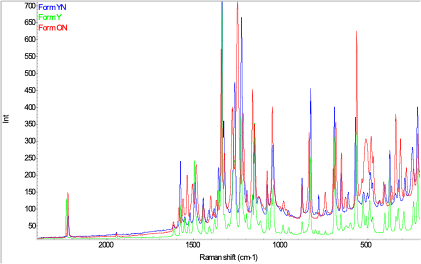
Figure 1: Raman microscopy spectra of different forms of ROY grown on a microscope slide

Figure 2: Yellow needles grown on a microscope slide (Form Y)
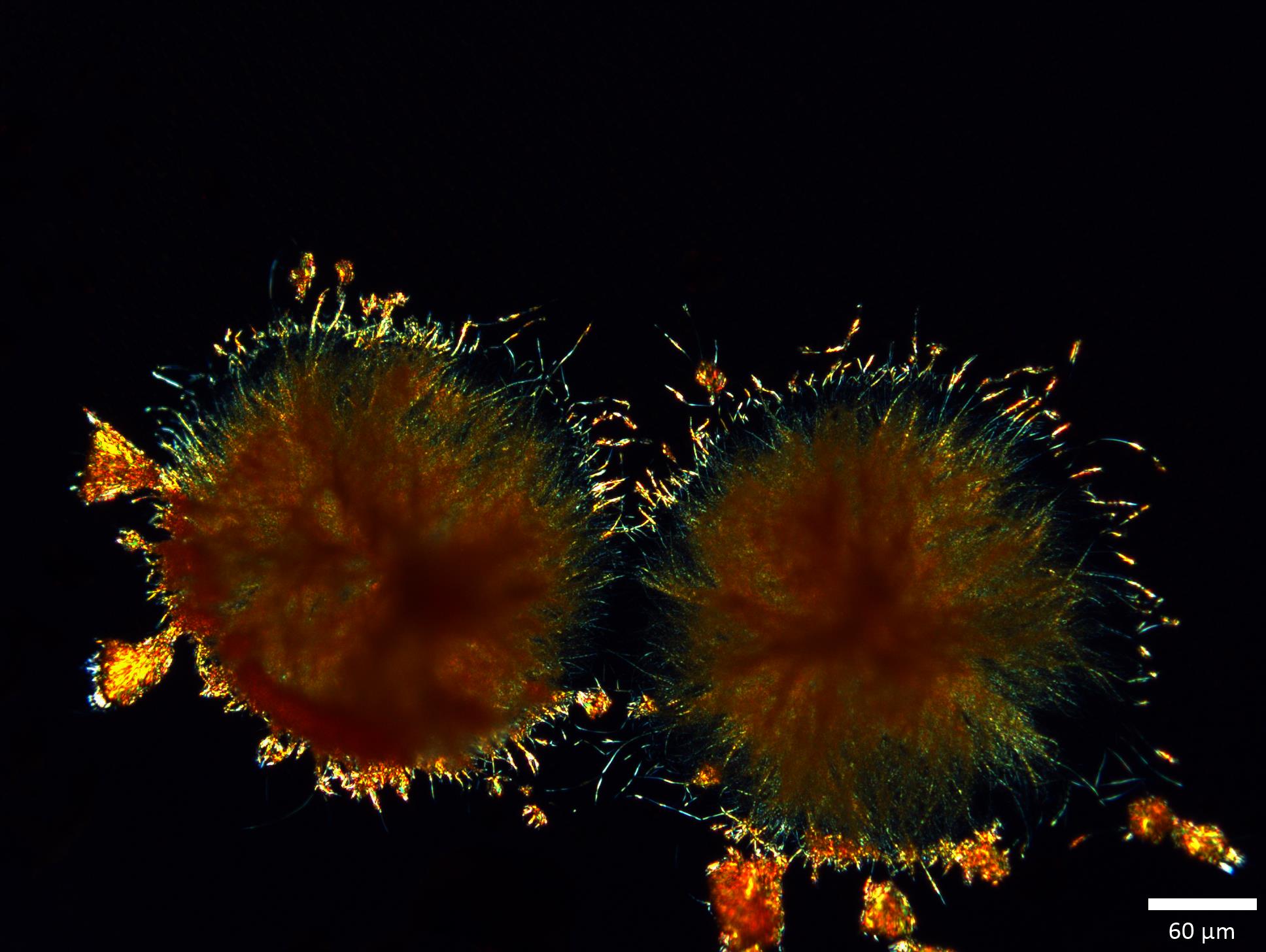
Figure 3: Orange rosettes grown on a microscope slide (Form ON)

Figure 4: Yellow blades grown on a microscope slide (Form YN)
