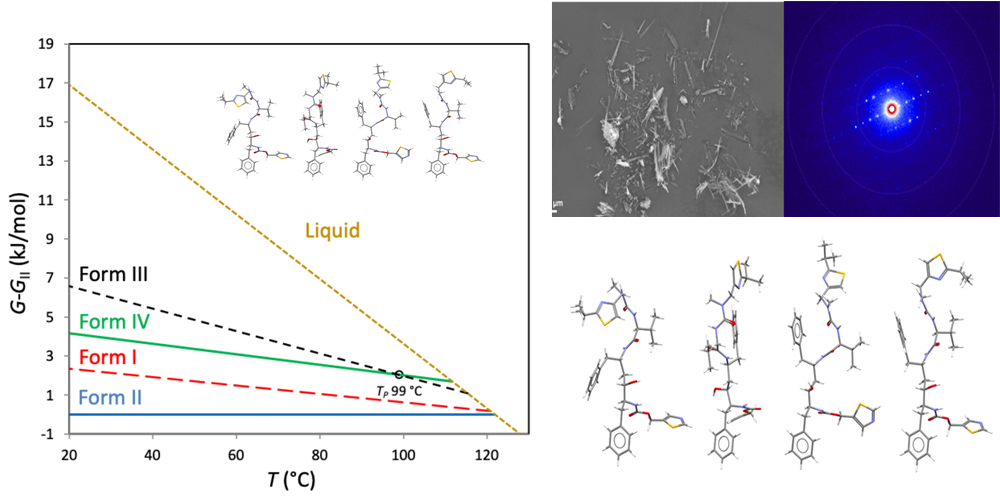
Improved Pharma Scientists to Present at Spring Pharmaceutical Synchrotron-XRPD Workshop May 6-8 in Basel, Switzerland
Ed olimpio2026-04-21T12:45:20+00:00Using Orthogonal Techniques to Probe for Detectable Crystalline API in Amorphous Dispersions WEST LAFAYETTE, Ind., April 21, 2026 /PRNewswire/ -- Detecting crystalline domains in amorphous materials can be critical for shelf-life stability of pharmaceuticals. Read more